|
9/26/2023 0 Comments Draw lewis structure for sf2
That was all about is SF4 polar or nonpolar. SF4 is heavier than air, and it reacts with water vigorously (with force and energy). The SF4 have colorless gas, which has a very distinct smell, This gas is very toxic, and it’s irritant to eyes and skin. Yes, SF4 can be hydrolyzed because, in SF4, the sulfur has a +4 oxidation state, but when they accept electrons pairs from H2O molecules, the oxidation state becomes +6. The SF4 has a see-saw structure, which leads to an overall dipole. 
Sulfur tetrafluoride (SF4) is an example of a seesaw shaped. Two are arranged in the same plan (axial), and the remaining two are equatorial. These four atoms are arranged in a different plane. In the seesaw shape, the central atom is surrounded by four adjacent atoms. The SF4 has a stronger dipole-dipole intermolecular force. 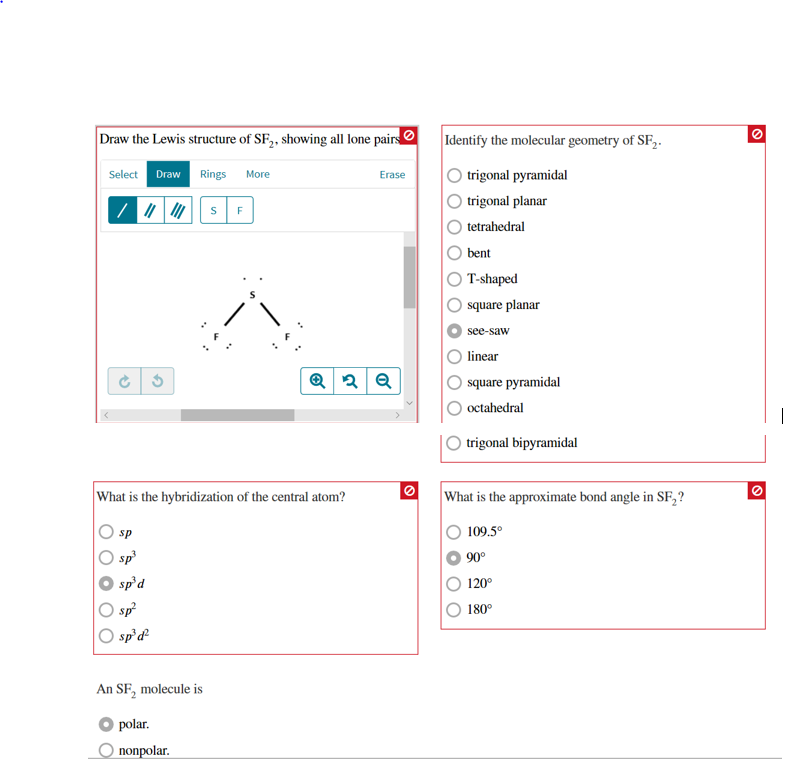
The above is the few properties of sulfur tetrafluoride (SF4) Sulfur Tetrafluoride Intermolecular Forces
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
